Unveiling the Microscopic Universe: The Intricate Chemical Symphony Behind Every Cup of Coffee

The journey from bean to brew is a complex dance of physical parameters, but the true magic of coffee unfolds at the molecular level, revealing a captivating world far beyond its seemingly simple appearance. While the vast majority—over 98.5%—of a typical cup of black coffee is water, the remaining 1.2-1.5% of total dissolved solids (TDS) represents a microscopic universe of hundreds, if not thousands, of distinct chemical compounds. These extracted solids, typically 12-15 grams per liter of brewed coffee from approximately 60 grams of grounds, are the architects of coffee’s renowned color, taste, and intoxicating aroma, challenging scientists and connoisseurs alike to fully comprehend its profound complexity.
The Quest to Define Coffee’s Essence
For centuries, coffee’s allure has captivated cultures worldwide, driving an industry worth hundreds of billions of dollars annually. Yet, the scientific endeavor to precisely map its chemical fingerprint is a relatively modern pursuit, gaining significant momentum with advancements in analytical chemistry. Early empirical methods of brewing have slowly given way to a more nuanced understanding of how specific chemical interactions create the sensory experience we cherish. The sheer number of molecules present in a single cup—many in concentrations so minute they defy easy detection—makes coffee one of the most chemically complex beverages known.
Chromatographic techniques, such as Gas Chromatography-Mass Spectrometry (GC-MS) and Aroma Extract Dilution Analysis (AEDA), have been instrumental in peeling back these layers. These powerful tools allow researchers to separate individual chemical components, revealing an astonishing array of compounds. However, the scientific community continues to grapple with identifying all the contributors to coffee’s distinct profile. A landmark AEDA study from 2002, for instance, reported that a significant portion—13 out of 40 identified key odorants in coffee—remained chemically unknown [1]. This highlights the persistent challenge: many crucial flavor molecules exist in extremely low concentrations, making their isolation and structural elucidation a formidable task. Additionally, some molecules might be large and structurally intricate, further complicating their characterization.
The difficulty in fully replicating coffee’s flavor underscores this complexity. A 1996 study demonstrated that synthetically concocting "model coffees" using 22 key volatile compounds could produce beverages described as "clearly coffee-like" by assessors, successfully mimicking Arabica and Robusta profiles [2]. While an impressive feat, the descriptor "clearly coffee-like" rather than "indistinguishable from natural coffee" eloquently testifies to the hundreds, if not thousands, of other compounds contributing to the authentic, multifaceted sensory experience that a simple model cannot yet capture. This ongoing scientific quest not only deepens our appreciation for coffee but also informs innovation in roasting, brewing technology, and sustainable agricultural practices.
The Dual Nature: Volatiles for Aroma, Non-Volatiles for Taste
The rich sensory profile of coffee is ingeniously divided between two primary categories of compounds: volatile and non-volatile. This fundamental distinction dictates how we perceive coffee, engaging both our olfactory and gustatory systems in a synchronized sensory symphony.
Volatile Compounds: The Architects of Aroma
Volatile compounds are those that readily evaporate at ambient temperatures, making their way into our nasal passages. These molecules, often present in incredibly low concentrations, are responsible for coffee’s complex and diverse aroma profile, which can range from floral and fruity to nutty, chocolatey, earthy, and even smoky. The human nose, an exquisitely sensitive analytical detector, can distinguish between thousands of different volatile compounds, each contributing a unique nuance to the overall bouquet. Pyrazines, for example, contribute to roasted and nutty notes, while furans often impart caramel or sweet undertones. Thiols can add desirable sulfurous notes, reminiscent of roasted meat or toast, in carefully balanced quantities. The interplay of these countless volatile molecules creates the distinctive aromatic signature of any given coffee, heavily influenced by bean origin, processing, and roast level.
Non-Volatile Compounds: The Foundation of Taste and Body
In contrast, non-volatile compounds remain largely in the liquid phase and are primarily responsible for the tastes perceived on the tongue, as well as the coffee’s body and texture. These compounds interact with taste receptors to produce the fundamental taste sensations:
- Bitterness: Primarily driven by chlorogenic acid lactones and melanoidins (products of the Maillard reaction during roasting), and, of course, caffeine. While often perceived negatively in excess, a balanced bitterness is crucial for coffee’s characteristic depth.
- Sweetness: Primarily from residual sugars (carbohydrates like sucrose and fructose) that survive the roasting process, and complex oligosaccharides.
- Acidity: Contributed by a variety of organic acids such as citric acid (often found in African coffees, imparting bright, fruity notes), malic acid (green apple notes), quinic acid (can contribute to perceived astringency and bitterness), and acetic acid (vinegar-like notes, typically in lower concentrations). Acidity provides brightness and vibrancy, preventing the coffee from tasting flat.
- Astringency: Often caused by phenolic compounds, particularly tannins and chlorogenic acids, which create a drying sensation in the mouth.
Beyond taste, non-volatile components significantly influence coffee’s body and texture. Lipids (oils) and insoluble fine particles, particularly prevalent in unfiltered coffees like French press or espresso, contribute to the perceived richness, viscosity, and mouthfeel. These elements create a sensation of fullness and weight on the palate, adding another dimension to the overall sensory experience. The intricate balance between these volatile and non-volatile components is what makes a cup of coffee far more than just a simple beverage—it is a finely tuned chemical symphony.
The Enigma of Trace Compounds: Where Parts Per Trillion Dictate Flavor
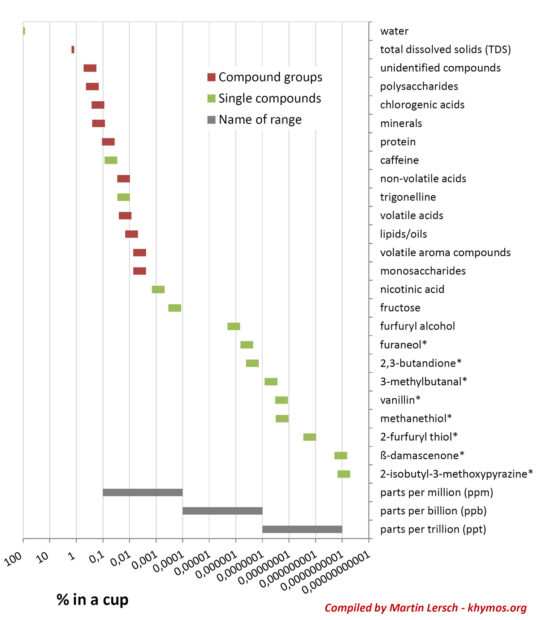
One of the most profound revelations in coffee chemistry is the disproportionate impact of compounds present in astonishingly low concentrations. While major components like water, caffeine, and carbohydrates dominate the mass percentage, many of coffee’s defining aromatic and gustatory characteristics are dictated by molecules measured in parts per million (ppm), parts per billion (ppb), or even parts per trillion (ppt). To visually comprehend this vast disparity, scientists often plot concentration ranges on a logarithmic scale, spanning more than nine orders of magnitude, highlighting the extraordinary breadth of coffee’s chemical landscape.
The key to understanding this phenomenon lies in the concept of Odor Activity Value (OAV). An OAV is calculated by dividing a compound’s concentration by its odor detection threshold—the minimum concentration at which a compound can be smelled. A high OAV indicates a strong sensory impact, even if the compound is present in minuscule amounts. This means that a compound present at 0.0000000001% (1 part per trillion) can be just as crucial to the overall flavor profile as another compound present at 0.01%, simply because our olfactory receptors are exquisitely sensitive to the former. This principle underscores why the human nose remains an unparalleled analytical detector, capable of discerning nuances that even the most sophisticated laboratory equipment might struggle to quantify in a meaningful sensory context.

Beta-Damascenone: A Potent Player from Rose Gardens to Coffee Cups
Among the pantheon of potent odorants in coffee, beta-damascenone stands out as a prime example of a trace compound with immense sensory impact. This remarkable molecule, a member of the rose ketone family, has been identified as one of the top five most impactful odorants in coffee, despite its presence in concentrations as low as 1 part per trillion (ppt) [1].
The story of beta-damascenone begins not in a coffee plantation, but in a rose garden. First discovered in rose oil in the 1960s, its name is derived from the Damask rose ( Rosa × damascena ), a flower celebrated for its intense fragrance. Its initial fame came in the perfumery world, where the Swiss fragrance and flavor company Firmenich pioneered its use. Beta-damascenone, along with other rose ketones, became a cornerstone ingredient in iconic perfumes, most notably Dior’s "Poison," launched in 1985. The molecule’s complex and multifaceted aroma profile—described by The Good Scents Company as a blend of fruity (apple, blackcurrant, plum), sweet, floral (rose), honey, tobacco, woody, rum, wine, and tea notes—made it a versatile and indispensable component in creating rich, lingering scents.
Its discovery as a pivotal compound in coffee further cemented its status as a critical flavor ingredient. The presence of such a versatile and potent molecule, bridging the aromatic worlds of roses and roasted coffee, illustrates the unexpected chemical connections that contribute to complex flavor profiles. Scientists continue to refine methods for accurately measuring odor thresholds and quantifying compounds at such ultralow concentrations, suggesting that further key odorants, yet to be fully characterized, may still be discovered in coffee in the years to come, further enriching our understanding of its sensory depth.
Extraction Science: Water’s Strengths and Limitations
The brewing process is fundamentally an act of extraction, where water acts as a solvent, drawing out soluble compounds from ground coffee. Understanding the differing solubility of various compounds in water is crucial for optimizing extraction and, consequently, the final flavor profile. Coffee compounds can be broadly categorized into polar and non-polar types, each interacting with water differently.
Polar Compounds: Water’s Forte
Polar compounds, which include acids, minerals, proteins, and carbohydrates (sugars and soluble oligosaccharides), share a common characteristic: they are highly soluble in water. As water itself is a polar solvent, it efficiently dissolves and extracts these components. Typically, over 75% of the polar compounds present in coffee grounds are extracted during a standard brewing cycle [2]. These compounds contribute significantly to coffee’s taste (acidity, sweetness, bitterness) and body, forming the backbone of the beverage’s perceived strength and foundational characteristics.
Non-Polar Compounds: The Challenge for Water
Conversely, non-polar compounds, which primarily encompass volatile aroma compounds and lipids (oils), have low solubility in water. As a result, water is far less efficient at extracting these components, typically dissolving only 10-30% of the non-polar compounds present in the grounds. This selective extraction has profound implications for coffee’s aroma and overall mouthfeel. The lipids, for instance, are responsible for the creamy texture and richness in beverages like espresso, where high pressure and fine grind facilitate their emulsification.
This disparity in extraction yields between polar and non-polar compounds is a critical factor in brewing. Different brewing methods inherently favor the extraction of certain compound types over others. For example, methods with longer contact times or higher temperatures may extract more of the less soluble compounds, potentially leading to over-extraction of bitter non-polar compounds. Conversely, rapid extractions, like espresso, rely on pressure to compensate for short contact times, extracting a unique balance.
This understanding also opens doors for experimental exploration. The possibility of using less polar solvents, such as ethanol, for coffee extraction suggests avenues for creating novel coffee products or highly concentrated flavor essences. Such approaches could unlock additional dimensions of coffee’s flavor profile, yielding extracts with vastly different chemical compositions and sensory characteristics than those achieved with water alone.
Beyond Total Dissolved Solids: The Human Palate Reigns Supreme
Total Dissolved Solids (TDS) is a widely used metric in the coffee industry, offering a quantifiable measure of coffee strength. It provides a valuable benchmark for consistency, allowing brewers to replicate recipes and ensure a predictable level of concentration. However, relying solely on TDS to gauge coffee quality or predict its flavor and aroma profile is a significant oversimplification.
While TDS quantifies the total mass of dissolved material, it offers no insight into the identity or sensory impact of those compounds. The compounds that contribute most significantly to TDS—primarily carbohydrates, acids, and caffeine—are not necessarily the same compounds that dictate the delicate nuances of aroma and flavor. As established, many of the most impactful aroma compounds exist in concentrations far too low to noticeably influence the TDS reading, yet they define the coffee’s aromatic signature.
Furthermore, the significant difference in extraction yields between polar (high solubility, contribute to TDS) and non-polar (low solubility, critical for aroma) compounds means that a TDS measurement is inherently skewed. It primarily reflects the extraction of water-soluble polar components and is a poor indicator of the presence or balance of the crucial, albeit less soluble, non-polar aroma compounds. For instance, two coffees could have identical TDS readings but possess entirely different flavor profiles due to variations in their trace volatile compounds or the specific balance of their non-volatile acids and sugars.
This fundamental limitation reinforces the irreplaceable role of human sensory evaluation. The sophisticated "analytical detector" that is the human nose and palate remains the ultimate arbiter of coffee quality. Techniques like cupping, where trained professionals systematically evaluate coffee’s aroma, taste, body, and aftertaste, are indispensable for assessing the true complexity and desirability of a brew. As we continue to explore the wonders of coffee extraction, scientific measurements will undoubtedly become more precise, but the nuanced, holistic judgment of human perception will always be the final, most crucial step in appreciating the profound sensory experience of coffee.
The Future of Coffee Chemistry: Innovation and Appreciation
The ongoing scientific exploration into coffee’s chemical makeup promises exciting advancements across the entire coffee value chain. For growers, a deeper understanding of specific compound precursors can inform breeding programs, leading to varieties with enhanced flavor profiles or greater resilience. For roasters, this knowledge refines the art and science of roasting, allowing for more precise control over the development of desired aromatic and taste compounds while minimizing undesirable ones. Equipment manufacturers can design brewing devices that optimize the extraction of specific compound groups, catering to diverse preferences.
Ultimately, this wealth of chemical data translates into a richer, more consistent, and more diverse coffee experience for consumers. It empowers enthusiasts to move beyond generic descriptors and appreciate the intricate molecular symphony playing out in every cup. As research continues to uncover the secrets held within that mere 1.5% of dissolved solids, the future of coffee promises not only innovative products and processes but also a profound and ever-deepening appreciation for this extraordinary beverage.
References
[1] Sanz, C.; Czerny, M.; Cid, C.; Schieberle, P. Eur. Food. Res. Technol., 2002, 214, 299 (DOI: 10.1007/s00217-001-0459-9)
[2] Semmelroch, P.; Grosch, W. J. Agric. Food Chem. 1996, 44, 537 (DOI: 10.1021/jf9505988)
[3] Belitz, H.-D.; Grosch, W.; Schieberle, P. (ed.), 2004, Food Chemistry (3rd edition) Springer.
[4] Amanpour, A.; Selli, S. J. Food Proc. Pres. 2016, 40, 1116 (DOI: 10.1111/jfpp.12692)
[5] Batali, M. E.; Frost, S. C.; Lebrilla, C. B.; Ristenpart, W. D.; Guinard, J.-X. J. Sci. Food Agric. 2020, 7, 2953 (DOI: 10.1002/jsfa.10323)