Unraveling the Chemical Symphony: What Truly Defines a Cup of Coffee?

The daily ritual of coffee brewing, a cherished practice across cultures, culminates in a cup that is far more complex than its seemingly simple appearance suggests. While consumers meticulously select beans and fine-tune brewing parameters—from grind size and water temperature to brew ratio and filter material—the true marvel lies in the intricate chemical tapestry woven within the liquid itself. This deep dive moves beyond the mechanics of extraction to explore the molecular universe residing within a typical black coffee, revealing the scientific basis of its captivating aroma, rich taste, and unique body.
Beyond the Bean: Deconstructing Coffee’s Core Components
At its most fundamental, a cup of black coffee is overwhelmingly composed of water, typically exceeding 98.5% of the beverage’s total volume. The remaining fraction, a mere 1.2% to 1.5%, constitutes the total dissolved solids (TDS). This seemingly small percentage translates to a substantial amount of extracted material: approximately 12 to 15 grams of soluble compounds derived from about 60 grams of coffee grounds per liter of brewed coffee. These extracted solids are the architects of coffee’s sensory profile, imparting its characteristic color, taste, and aroma.
The journey from bean to brew involves a remarkable transformation, where roasting unlocks precursors and brewing solubilizes a vast array of compounds. The composition is a dynamic interplay, influenced not just by the bean’s origin and processing but also by every decision made during preparation. Understanding these dissolved solids is crucial for both scientific inquiry and the pursuit of the perfect cup.
A Symphony of Molecules: The Intricate World of Coffee Aroma and Taste
Analytical chemists, utilizing sophisticated chromatographic techniques, have long sought to catalogue the full spectrum of molecules present in coffee. What they consistently discover is a staggering number: hundreds, if not thousands, of individual chemical components. While many of these have been identified and characterized, a significant portion continues to elude precise identification, posing an ongoing challenge for researchers.
Early investigations, such as an aroma extract dilution analysis (AEDA) conducted in 2002, underscored this complexity, revealing that 13 out of 40 key odorants identified in coffee remained unknown. The difficulty in characterizing these elusive compounds stems from several factors. Often, they are present in extraordinarily low concentrations, making it challenging to isolate sufficient quantities for full structural elucidation using techniques like Nuclear Magnetic Resonance (NMR) or Mass Spectrometry (MS). Furthermore, some molecules may possess large, complex structures, complicating the process of resolving their exact chemical makeup.
The quest to artificially replicate coffee flavor highlights this complexity. A pioneering study in 1996 demonstrated that even with a carefully selected panel of 22 key volatile compounds, synthetic "model" coffees, mimicking Arabica and Robusta, were merely described as "clearly coffee-like" by sensory assessors. This suggests that a truly authentic coffee experience requires a far more intricate blend of compounds, many of which remain unidentified or are present in concentrations previously considered negligible. The sheer number and synergistic interactions of these molecules contribute to the inimitable depth and nuance of natural coffee.
The Elusive Odorants: A Scientific Pursuit
The scientific community continues to invest heavily in unraveling coffee’s chemical secrets. Advances in high-resolution mass spectrometry and gas chromatography-olfactometry (GC-O) are providing unprecedented insights into the volatile fraction. Researchers are identifying new pyrroles, pyrazines, furans, and thiophenes—classes of compounds known for their roasted, nutty, and savory notes—along with esters, aldehydes, and ketones contributing fruity and floral nuances. The "unknowns" often represent novel classes of compounds or highly reactive intermediates formed during roasting, demanding innovative analytical approaches. This ongoing research not only expands our understanding of coffee but also pushes the boundaries of flavor chemistry itself.
Volatile vs. Non-Volatile Compounds: The Dual Contribution to Flavor
The myriad compounds in coffee can be broadly categorized into two major groups based on their physical properties and sensory contributions: volatile and non-volatile compounds.
Volatile compounds are those that readily evaporate at room temperature, making their way into the olfactory system and contributing to coffee’s distinct aroma. These are primarily responsible for the "smell" experienced before and during consumption. Non-volatile compounds, conversely, are stable in solution and interact with taste receptors on the tongue, influencing the "taste" profile of the beverage. While some molecules can affect both aroma and taste, the human nose typically exhibits a much higher sensitivity (lower threshold) to volatile aroma compounds than the tongue does to non-volatile taste compounds.

The non-volatile components are responsible for the fundamental tastes perceived in coffee, including bitterness (often attributed to chlorogenic acid lactones and melanoidins), sweetness (from residual sugars and certain sugar alcohols), acidity (from various organic acids like quinic, citric, malic, and acetic acids), and astringency (polyphenols and tannins). Beyond taste, non-volatile compounds, particularly dissolved oils and tiny insoluble particles (micro-fines), significantly contribute to the "body" and "texture"—the mouthfeel—of coffee.
A closer look at the soluble components reveals a diverse chemical inventory:
- Carbohydrates: Sugars (e.g., sucrose, fructose) and soluble oligosaccharides provide sweetness and contribute to body.
- Acids: A complex blend of organic acids (chlorogenic acids, quinic acid, citric acid, malic acid, acetic acid, etc.) that define coffee’s brightness, tartness, and can also contribute to bitterness.
- Minerals/Salts: Trace elements that influence water chemistry and can subtly affect taste.
- Proteins: While many proteins are denatured during roasting, some soluble peptides and amino acids contribute to flavor precursors and body.
- Lipids: Small amounts of emulsified oils contribute to body, richness, and can carry volatile aroma compounds.
- Caffeine: The well-known stimulant, contributing a distinct bitterness.
- Volatile Aroma Compounds: The hundreds of molecules responsible for the intricate bouquet.
- Unidentified Compounds: A substantial fraction, likely contributing to color, bitterness, and overall complexity. These often include large polymeric structures like melanoidins, formed during the Maillard reaction, which are critical for coffee’s characteristic brown color and perceived body.
The Power of Perception: How Concentration Deceives
One of the most profound insights into coffee chemistry is the realization that a compound’s concentration does not directly correlate with its sensory impact. The human olfactory system possesses an astonishing ability to detect certain molecules at incredibly low levels, thanks to unique odor thresholds for each compound. This phenomenon is quantified by the concept of the Odor Activity Value (OAV), defined as the ratio of a compound’s concentration to its odor threshold. A high OAV indicates a significant contribution to the overall aroma, even if the compound is present in minute quantities.
Consider the logarithmic scale used to illustrate the concentration ranges of coffee compounds, spanning more than nine orders of magnitude. While water and bulk dissolved solids occupy the highest percentages, the impact of trace compounds cannot be underestimated. A compound present at an astonishingly low concentration, say 0.0000000001% (equivalent to 1 part per trillion, or ppt), can be as crucial to the overall flavor profile as another compound present at 0.01% (100 parts per million, or ppm), simply because our nose is exquisitely sensitive to the former. This intricate interplay underscores why "synthetic" coffee remains a distant prospect; replicating such a vast and varied spectrum of sensitivities is an enormous challenge.
Beta-Damascenone: A Potent Player from Petals to Brew
Among the pantheon of potent odorants, $beta$-damascenone stands out as a prime example of a compound with disproportionate impact relative to its minuscule concentration. Found in coffee at levels as low as 1 part per trillion (ppt), this molecule is nevertheless considered one of the top five most impactful odorants in coffee. Its contribution is so significant that it can be a decisive factor in the overall perceived flavor.
$beta$-damascenone belongs to a family of compounds known as rose ketones, first identified in rose oil during the 1960s. Its name is derived from the Damask rose (Rosa × damascena), a flower renowned for its exquisite fragrance. The discovery of rose ketones revolutionized the perfumery industry, with flavor and fragrance powerhouse Firmenich pioneering their use. $beta$-damascenone, in concert with other rose ketones, famously played a central role in the iconic Dior perfume, Poison, showcasing its potent and versatile aromatic qualities.
The sensory profile of $beta$-damascenone is remarkably complex and multifaceted. The Good Scents Company, a leading resource in the flavor and fragrance industry, lists its odor descriptors as fruity (apple, blackcurrant, plum), floral (rose, violet, honey), woody, tobacco-like, and even some spicy or savory undertones. This broad spectrum of aromatic notes allows $beta$-damascenone to weave seamlessly into the diverse tapestry of coffee aroma, enhancing its fruitiness, adding depth to its floral character, and contributing to its overall complexity.
From Rose Gardens to Coffee Cups: A Chronology of Discovery
The journey of $beta$-damascenone from its initial isolation in rose oil to its identification as a key coffee odorant highlights the evolution of analytical chemistry. The 1960s saw the rise of advanced chromatography techniques, which enabled the separation and identification of complex mixtures in natural products. Its application in high-end perfumery in the subsequent decades cemented its status as a high-impact aroma chemical. Later, with the refinement of gas chromatography-mass spectrometry (GC-MS) coupled with olfactometry (GC-O), researchers in the 1990s and early 2000s were able to systematically identify and quantify trace aroma compounds in roasted coffee. This led to the groundbreaking discovery of $beta$-damascenone’s crucial role, despite its minute presence. The excitement within the scientific community was palpable, as it reinforced the understanding that coffee’s magic resided not just in its abundant constituents but in the delicate balance of these powerful trace compounds.
Accurately measuring odor thresholds and quantifying compounds at such ultralow concentrations requires meticulous and highly specialized laboratory work. Given these analytical challenges, it is entirely plausible that additional, equally potent, and previously undiscovered key odorants in coffee await identification in the coming years, continuing to deepen our appreciation for this complex beverage.
The Art of Extraction: Water’s Selective Embrace
The brewing process is fundamentally an act of selective extraction, where water acts as a solvent to draw out soluble compounds from ground coffee. The efficiency and selectivity of this extraction are largely determined by the polarity of the compounds.
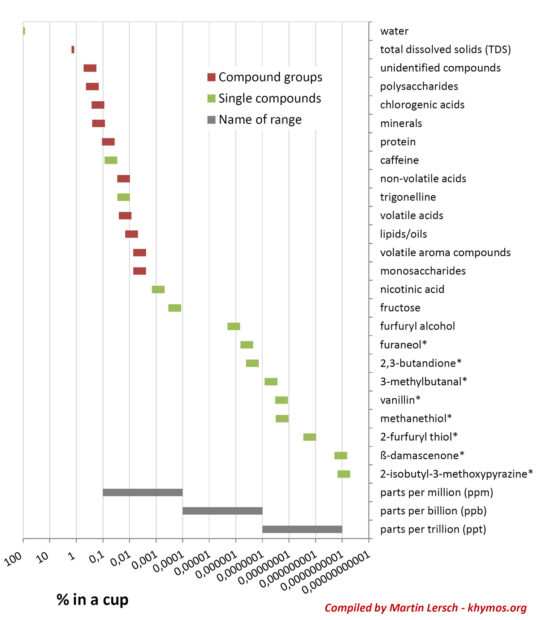
Coffee’s chemical constituents can also be broadly classified as polar and non-polar compounds.
- Polar Compounds: This group includes acids, minerals, many proteins, and carbohydrates (sugars, oligosaccharides). These compounds are highly soluble in water due to water’s polar nature and its ability to form hydrogen bonds. Consequently, when brewing coffee with water, typically more than 75% of these polar compounds are effectively extracted. These contribute significantly to the perceived strength, acidity, sweetness, and overall body of the coffee.
- Non-Polar Compounds: This category encompasses most volatile aroma compounds and lipids (oils). These molecules have low solubility in water. As a result, only a small fraction, typically between 10% and 30%, of the non-polar compounds are extracted during water-based brewing. This selective extraction means that a significant portion of the lipid-soluble flavor precursors and oils remain trapped within the spent coffee grounds.
This fundamental difference in extraction efficiency has profound implications for brewing. Water, while an excellent solvent for polar compounds, is a relatively poor solvent for many of the crucial non-polar aroma compounds. This explains why varying brewing parameters, such as water temperature, contact time, and grind size, can dramatically alter the final flavor profile. For instance, hot brewing extracts more soluble material overall, including some less desirable bitter compounds, while cold brewing, with its longer extraction time at lower temperatures, tends to yield a less acidic and less bitter concentrate, as certain bitter acids are less soluble in cold water. Espresso, with its high pressure and short contact time, rapidly extracts a concentrated brew rich in both polar and some emulsified non-polar compounds, contributing to its intense flavor and crema.
Optimizing the Brew: Science Meets Craft
Understanding this polar/non-polar dynamic is key for baristas and roasters striving for optimal flavor. It informs decisions about roast profiles (which affect compound formation and solubility), grind settings (surface area for extraction), and brew methods (temperature, time, turbulence). The challenge lies in extracting desirable compounds while minimizing the extraction of undesirable ones.
For industrial applications, the concept of alternative solvents has long been explored. For example, supercritical carbon dioxide (scCO2) or organic solvents like ethanol are used in decaffeination processes and in the production of coffee extracts for flavorings. These less polar solvents can selectively extract different sets of compounds, particularly a higher percentage of the non-polar volatile aroma compounds and lipids, offering pathways to develop novel coffee products or enhance existing ones. While not practical for home brewing, this highlights the potential beyond water’s limitations.
The Limits of Measurement: Why the Human Palate Reigns Supreme
Total Dissolved Solids (TDS) is a widely used metric in the coffee industry, primarily serving as an indicator of brew strength and consistency. It provides a quantifiable measure of how much material has been extracted from the coffee grounds into the water. However, while useful for quality control and ensuring repeatable brews, TDS measurements offer a limited perspective on the overall sensory experience.
As demonstrated by the vast range of compound concentrations, only a few major compound groups contribute significantly to the bulk TDS value. These are typically the highly soluble polar compounds like carbohydrates, acids, and minerals. The problem arises because there is no direct correlation between the extraction rate of these TDS-influencing compounds and the myriad of minute, yet flavor-critical, volatile aroma compounds. The large disparity in extraction yields between polar and non-polar compounds further underscores this disconnect. A brew with a "perfect" TDS reading might still be lacking in aromatic complexity if the extraction conditions were suboptimal for the non-polar aroma molecules.
Consequently, while a TDS measurement can tell you how strong your coffee is, it is remarkably poor at predicting the nuanced aroma and intricate flavor profile of a cup of coffee. It cannot capture the delicate balance of fruity notes, the depth of roasted flavors, or the subtle floral undertones that define a truly exceptional brew.
This limitation means that despite the advancements in analytical chemistry, the most sophisticated and irreplaceable analytical detector for coffee remains the human nose and palate. Sensory evaluation, through practices like cupping, where trained professionals systematically assess aroma, taste, body, acidity, and balance, continues to be the gold standard for evaluating coffee quality. It is this experimental, subjective, yet highly refined approach that allows us to truly explore and appreciate the wonders of coffee extraction and the boundless variations it can yield.
The Future of Flavor: Continuous Discovery and Innovation
The ongoing scientific exploration into coffee’s chemical makeup promises continued innovation across the industry. For specialty coffee, a deeper understanding of molecular interactions can inform roasting strategies to selectively enhance desirable precursors or optimize brewing methods to unlock specific flavor notes. For the broader food and beverage industry, insights into the "unknowns" and the mechanisms of potent odorants like $beta$-damascenone could lead to the development of more authentic and complex coffee-flavored products.
Furthermore, the integration of artificial intelligence and machine learning with advanced analytical techniques is beginning to create predictive models that correlate chemical profiles with sensory attributes, potentially revolutionizing how coffee quality is assessed and optimized. However, the inherent complexity of thousands of interacting molecules, each with its unique threshold and synergistic effects, ensures that the art of coffee tasting and the human element will remain central to appreciating this globally beloved beverage. The journey to fully understand what is in a cup of coffee is far from over, offering a rich and rewarding field for scientists, producers, and enthusiasts alike.